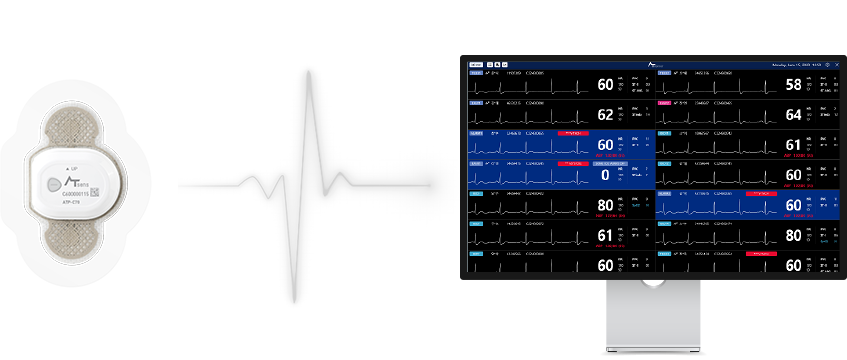
- AT-Patch
- AT-Note
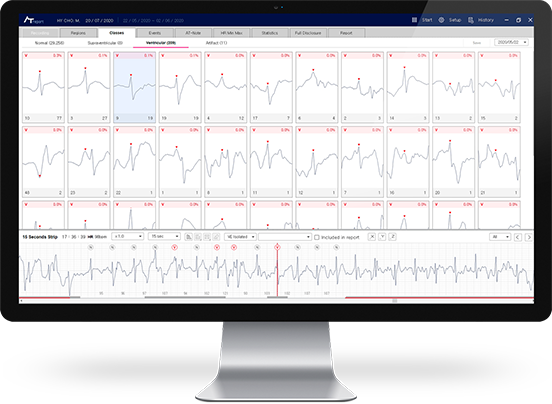
- AT-Report
- AT-Monitoring
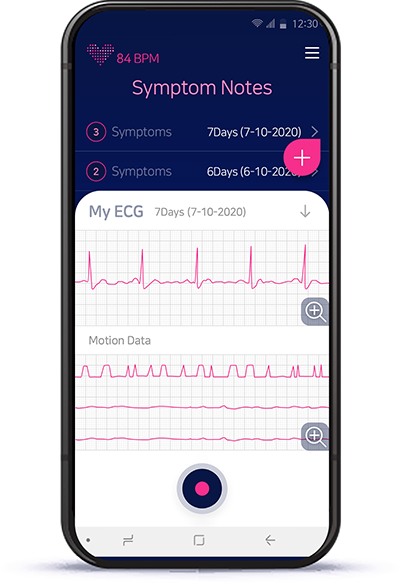
 Live ECG viewer via bluetoothAT-Note provides real-time ECG data and 3-Axis sensor(motion) data from AT-Patch.
If you have a compatible smartphone, it can easily be used.
Live ECG viewer via bluetoothAT-Note provides real-time ECG data and 3-Axis sensor(motion) data from AT-Patch.
If you have a compatible smartphone, it can easily be used.
 Easy to record your symptomThere are no need to carry pen or papers. When you feel any unusual heartbeats,
push the power-button shortly. You can simply note your symptoms on the app.
Easy to record your symptomThere are no need to carry pen or papers. When you feel any unusual heartbeats,
push the power-button shortly. You can simply note your symptoms on the app.
※ Without using the app, symptoms are recorded in the memory of the patch after pushing the power-button.
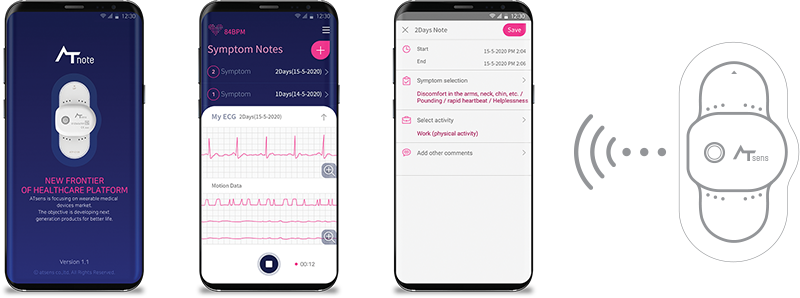
Symptom Note without Smart PhoneIf you are located in remote places or no compatible smartphone, you can note your symptom
on the ‘Symptom Note’ paper enclosed in the AT-Patch package.

* Disclaimer
1. The above information is based on the products, ATP-C130 obtained FDA, CE MDD(Medical Devices Directive), PMDA in Japan, MFDS in South Korea and ATP-C70 obtained PMDA in Japan, MFDS in South Korea. Thus, the information is specifically for South Korea & Europe and can differ with countries.
2. AT-Note(App) is usable only if you own a compatible smartphone or tablet. And the usage time of AT-Patch may be affected, depending on the individual communication environment for AT-Note’s Live ECG. For more information, please refer to the user manual.
3. AT-Report(PC S/W) is software for medical institutions, not provided patients directly. Patients can review the analysis result with or from your doctor.
4. The above information can be updated repeatedly.